predict which element forms the most stable compounds in the +2 oxidation state. Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. (See the above or below periodic table to see the valence electrons of inner transition metals). Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Another is when each of 3 p orbitals have one electron they start to pair as new ones are added (like when moving from nitrogen to oxygen). Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. One is that when electrons start to fill p orbital the ionization energy goes down a little. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements?
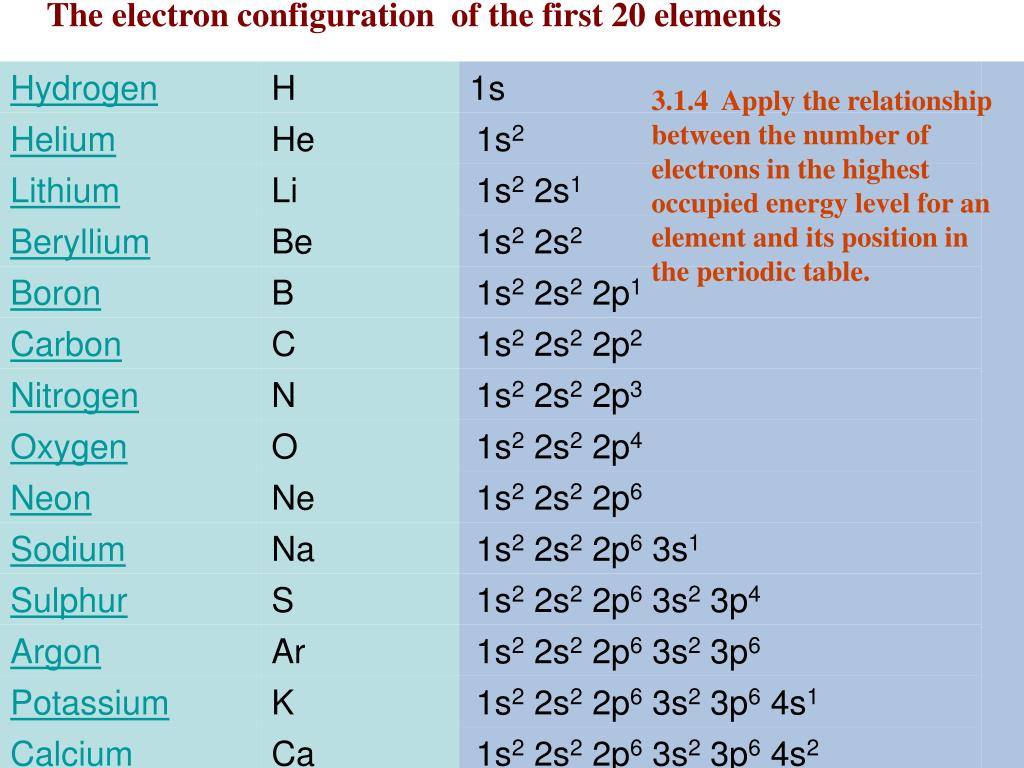
Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). The magnesium element has 2 electrons in outermost orbit.
